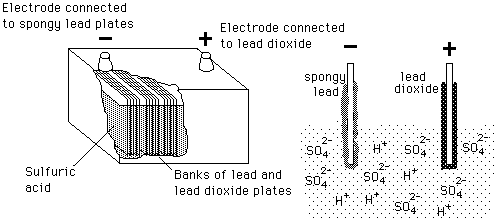
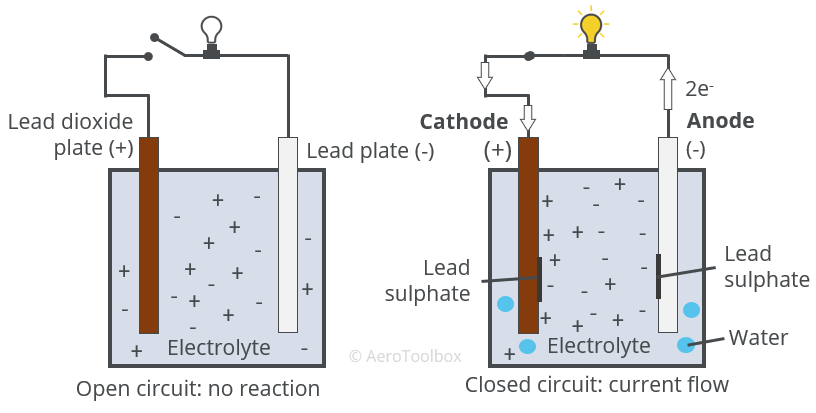
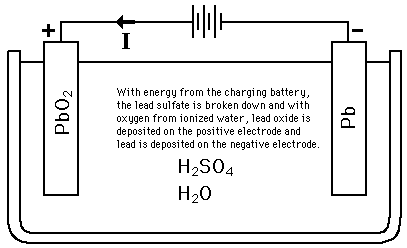
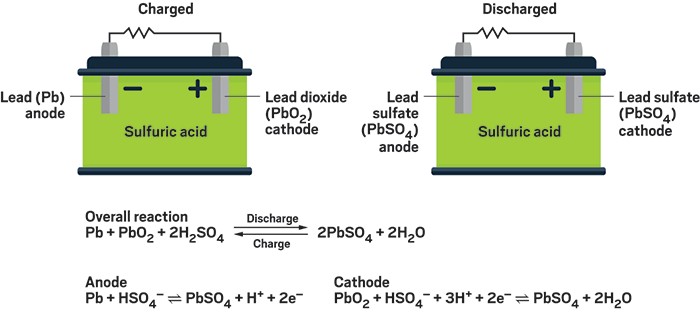
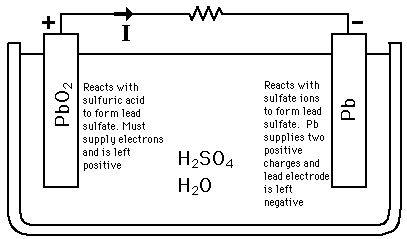
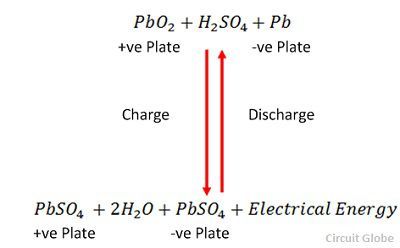
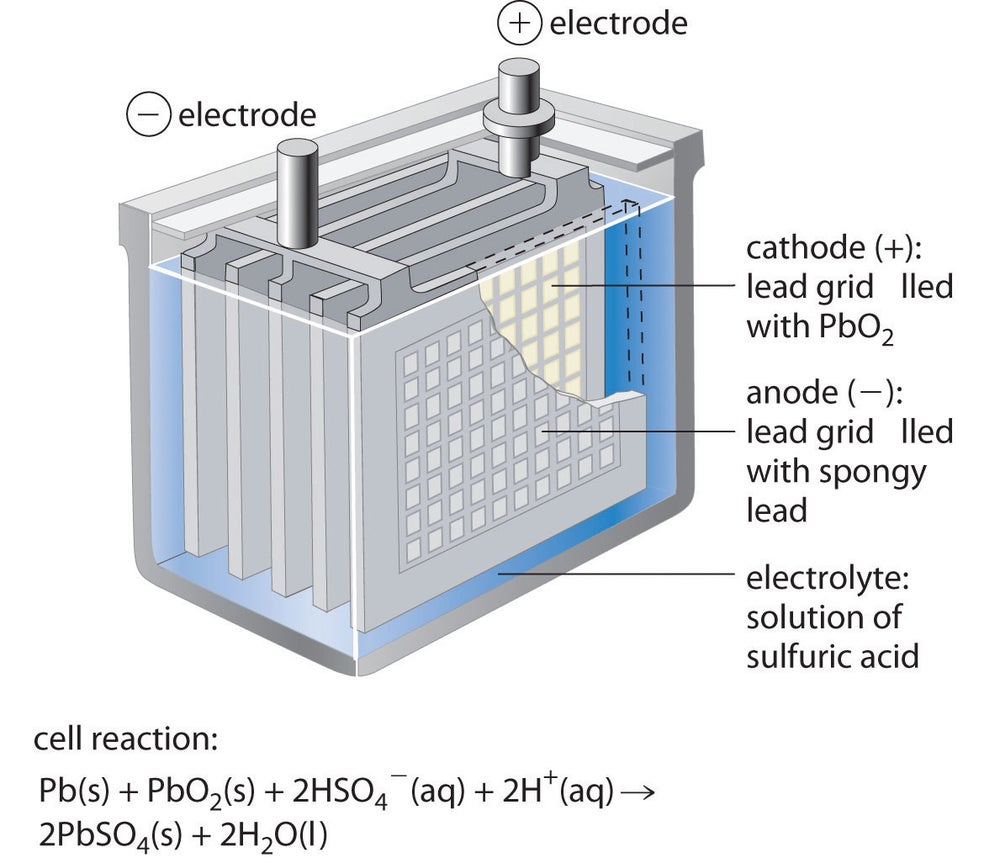
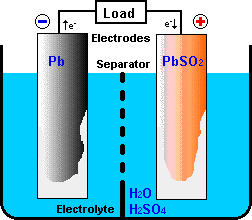
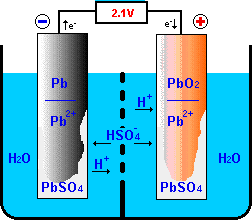
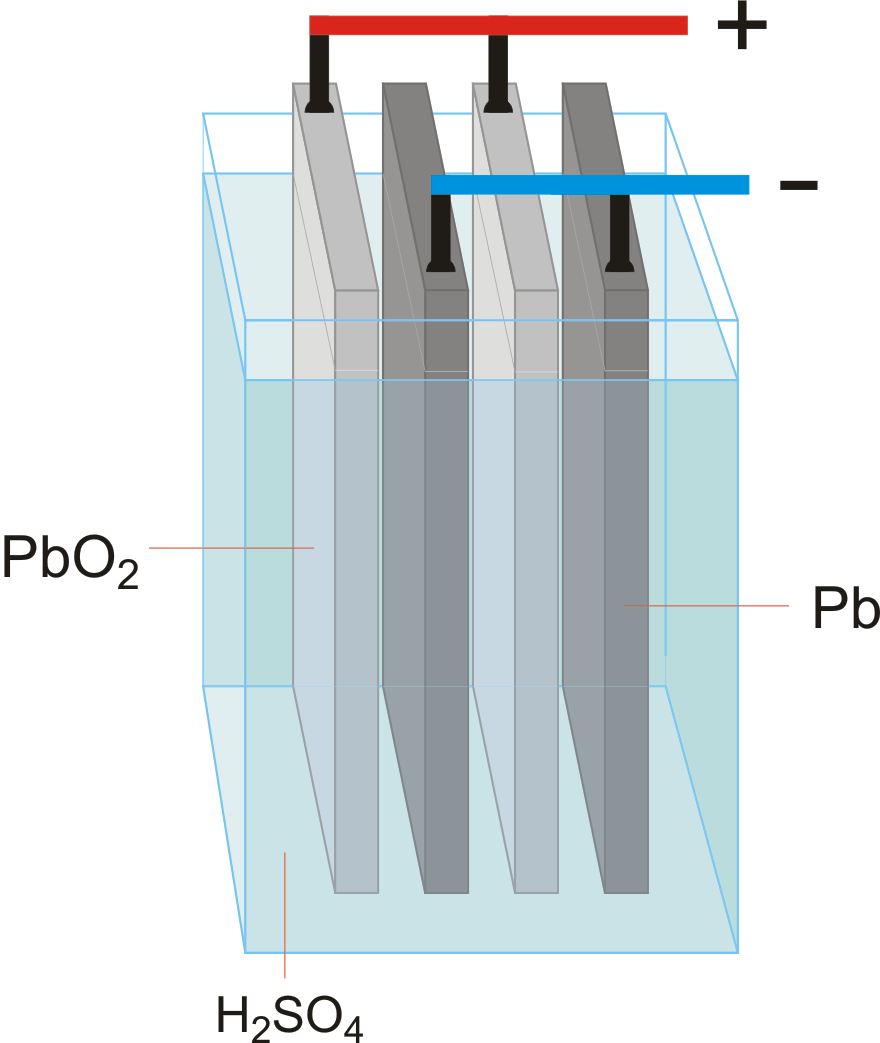
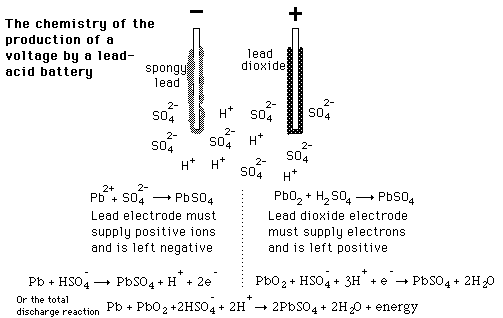The half reactions that occur in a lead acid battery are: `PbSO_(3)(s)+2e^(-)toPb(s)+SO_(4) - YouTube
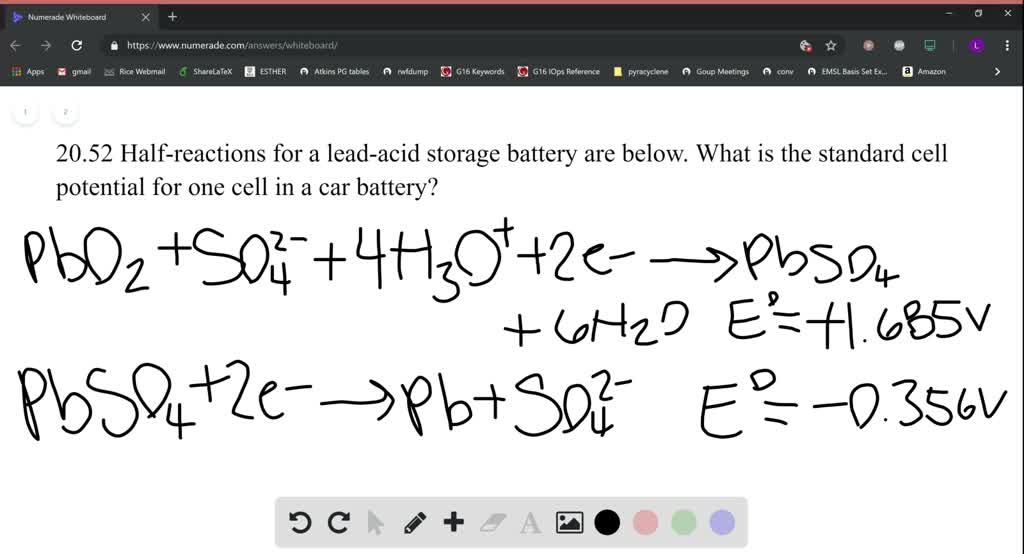
SOLVED:Half-reactions for a lead-acid storage battery are below. PbO2(s)+SO4^2-(aq)+4 H3O^+(aq)+2 e^-→ PbSO4(s)+6 H2O(l) E^0=+1.685 V PbSO4(s)+2 e^-→Pb(s)+SO4^2-(aq) E^0=-0.356 V What is the standard cell potential for one cell in a car battery?

SOLVED: Answer the four questions below regarding an electrochemical cell in an automotive lead-acid battery. The cell s anode made of lead and the cathode made of lead(IV) oxide Both are submerged